Both Regfo and Weave Bio use AI for regulatory compliance in biotech. That's about it for what they share.
Weave Bio writes your regulatory documents. Regfo checks whether your studies and protocols actually meet FDA requirements before you start writing anything. People keep asking me which one "wins" — well, not exactly. They solve different problems, and a Series A biotech trying to clear a first IND needs one of them much more than the other.
If you're a 10-person biotech trying to figure out whether your preclinical package has gaps before you spend another $200K on the next study, keep reading.
What's the difference between Regfo and Weave Bio in one sentence?
| Regfo | Weave Bio | |
|---|---|---|
| What it does | Checks protocols and study packages against FDA/ICH rules | Drafts regulatory dossiers (IND, eCTD) from source data |
| Core function | Compliance checker | Document authoring |
| Output | Gap analysis with specific ICH citations, compliance score 0-100 | Draft IND sections, tables, narratives in eCTD format |
| Time to value | Paste protocol, get findings in 30 seconds | Weeks of onboarding and data room setup |
| Pricing | $499/mo Starter, $1,499/mo Professional | Not published. Enterprise sales only. |
| Free trial | Yes | No |
| Target customer | Series A-C biotech, small CROs, 10-50 employees | Large pharma, enterprise CROs |
| Regulatory focus | FDA/ICH preclinical and clinical requirements | eCTD document structure and formatting |
| Founded | 2024 | 2022 |
| Funding | Pre-seed | $36M (Series A) |
What does Regfo actually do for preclinical compliance?
Regfo is a compliance checker. Upload your protocol or configure your study package, and it tells you what's missing according to FDA and ICH guidelines.
The rules engine covers 1,054 requirements across 24 ICH and FDA guidelines. When it finds a gap, it doesn't say "you might have an issue." It says: "ICH S7B Section 2.1 requires a hERG assay under GLP conditions. Your cardiovascular safety pharmacology package doesn't include one."
That's what your RA lead actually needs. Not a vague warning, but the exact ICH section and what's wrong.
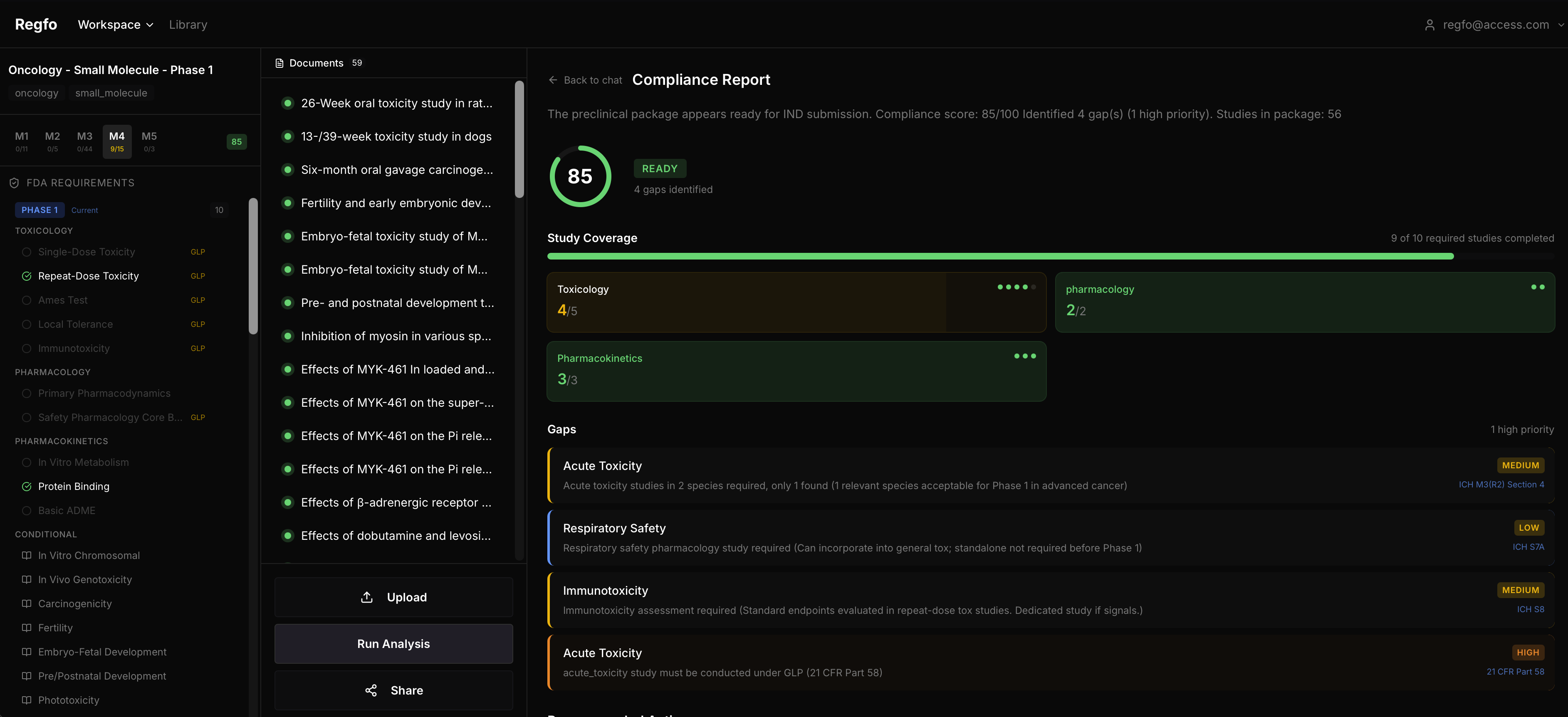
What you get:
- Compliance score 0-100 for your study package
- Specific findings with ICH/FDA guideline citations
- Gap analysis showing what studies are missing by phase
- AI narrative explaining each gap and how to fix it
- 25+ study types analyzed across Phase 1, 2, and 3
What you don't get:
- Document drafting. Regfo doesn't write your IND sections.
- eCTD formatting. We check compliance, not document structure.
- Post-submission tracking. We focus on pre-submission readiness.
Priced for small biotech teams: $499/mo to start, $1,499/mo for the full platform.
What does Weave Bio focus on?
Weave Bio is a document authoring platform. It helps regulatory teams draft IND applications and other eCTD submissions using AI.
Their flagship product, AutoIND, takes your source data (study reports, lab results, prior submissions) and generates draft regulatory documents: narratives, tables, figures, formatted to eCTD standards. Reviewers then edit and refine the drafts.
They've raised $36M through their Series A in October 2025, led by USVP. Advisory board includes people from Takeda, Gilead, and Boehringer Ingelheim. Parexel is their exclusive CRO partner.
What they do well:
- Document drafting with AI (IND sections, narratives, tables)
- Source traceability (AI outputs link back to source data)
- Collaborative review workflows
- Data Room for managing source files
- Enterprise-grade for large pharma teams
Where it falls short:
- No public pricing. You need a demo call to find out what it costs. This drives me crazy — it's 2026.
- Strongest for IND applications. NDA, BLA, and international (EMA, PMDA) support is still developing.
- No public reviews on G2, Capterra, or TrustRadius yet. (Worth noting: that might just mean they're still early, not that the product is bad.)
- Takes weeks to set up. Not a paste-and-go tool.
- Main proof point is one pilot with Takeda — 97% time savings on drafting tasks, but drafting is only part of the regulatory workflow.
Which tool is right for your stage?
This isn't about which is better. Different tools, different problems. Here's the honest breakdown.
Use Regfo when:
- You're writing or reviewing a protocol and need to know if it meets FDA requirements
- Your preclinical package might have gaps and you need to know which studies are missing
- You're a 10-50 person biotech and can't afford $100K/year enterprise tools
- You need specific ICH/FDA citations to justify compliance decisions
- You want answers in minutes, not weeks
Use Weave Bio when:
- You have a complete study package and need to draft the actual IND submission
- Your team has 50+ people and a dedicated regulatory writing group
- You need eCTD-formatted documents with automated table and figure generation
- You have budget for enterprise SaaS and time for onboarding
- You're working with a CRO like Parexel that has a Weave Bio license
Use both:
Check your study package with Regfo first. Find the gaps. Fix them. Then use Weave Bio (or your CRO, or your regulatory writing team) to draft the submission documents.
You check first, then write. One comes before the other.
How does pricing compare — Regfo vs Weave Bio?
Weave Bio doesn't publish pricing. Based on their enterprise positioning and Takeda/Gilead/Boehringer advisory board, expect six figures annually with multi-month implementation. (I've asked. They won't say. This is a pattern with enterprise regulatory SaaS.)
Regfo is transparent:
| Plan | Price | What you get |
|---|---|---|
| Starter | $499/mo | Protocol compliance checker, gap analysis, 25+ study types |
| Professional | $1,499/mo | Full platform, team collaboration, AI narratives, priority support |
| Enterprise | Custom | Dedicated instance, SSO, custom integrations |
Series A biotech. $200K/month burn. The difference between $499/mo today and an enterprise procurement cycle? Months.
Why did we build Regfo when Weave Bio already existed?
"I watched small biotech teams burn months because nobody caught a missing GLP-compliant tox study early enough. The protocol went through three amendment cycles. Each one cost 2-3 months. The problem wasn't document formatting. People didn't know what FDA actually required until it was too late."
— Vitaliy Zarubin, Founder of Regfo & The BrightByte
Weave Bio is solving document authoring for large pharma. That's a real problem worth solving. But it's not the problem that kills Series A biotechs. What kills them is submitting an IND and getting a clinical hold because they missed a requirement buried in an ICH guideline footnote.
That's what Regfo catches. 1,054 rules. 24 guidelines. Specific citations for every finding. In 30 seconds.
Check your protocol at regfo.com
Sources
- Weave Bio $36M Series A announcement — USVP — October 2025 funding round details
- Parexel and Weave Bio partnership announcement — exclusive CRO partnership
- ICH Guidelines portal — the 24 guidelines referenced by Regfo's rules engine
- FDA 21 CFR 312 — Investigational New Drug Application — U.S. regulatory baseline for IND submissions
- FDA Clinical Hold Statistics — Annual IND Review Report — context on why missed preclinical requirements matter